   
11 Additionally, as made clear by Bartoli and Todesco, 12 reactivity in these S NAr processes can be influenced by the size of the leaving group, by the basicity and size of the nucleophile, by interactions between nucleophile and leaving group, by the substrate framework (Miller’s term for the ring and its substituents), and by solvent effects, particularly on the nucleophile and leaving group. Also to be considered are the polarizabilities of both nucleophile and leaving group. However, the number of factors governing reactivity in S NAr reactions is large. This explanation, with references, is also given as one of several pertinent factors in Miller’s 1968 monograph on aromatic nucleophilic substitution. In some advanced organic texts one finds, unattributed, the statement that the greater polarity of the C–F bond allows an attraction between nucleophile and carbon, 8, 9 an attraction which is presumably considered to exist in the transition state (ts) of step 1 as well. 7Ī number of explanations have been given for the element effect. Where found it establishes the addition-elimination mechanism for S NAr reactions in which step 1, addition of the nucleophile to form a tetrahedral Meisenheimer complex (a substituted cyclohexadienide anion), is rate determining. 3 This result, called the “element effect” 7 is found for a number of nucleophilic types including oxy-anions, amines, and sulfur anions, and is especially prominent for smaller, less polarizable nucleophiles in protic solvents. 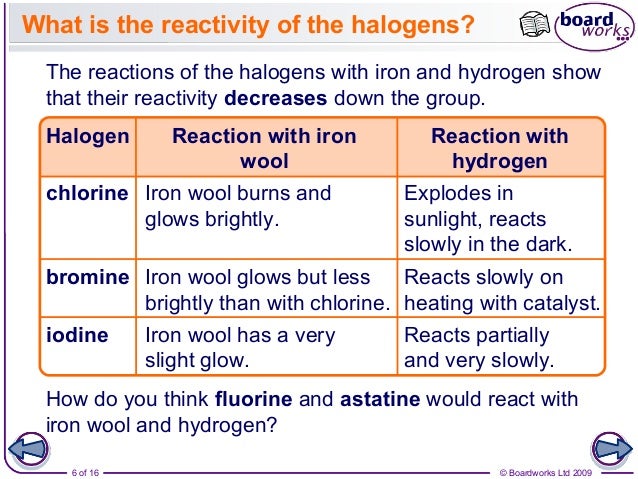
Bottom: The overall result for reaction of piperidine with 2,4-dinitrophenyl substrates.Ĭompared with S N2 reactions of aliphatic halides, an inverted order of halide leaving group abilities, F > Cl ≈ Br > I is often found in studies of rates of S NAr reactions of activated aromatic halides, e.g., substrates substituted at ortho and/or para positions on the ring with strong electron withdrawing groups (EWGs), commonly nitro, cyano, acyl, and even metal-substituted aryl halides in which vacant metal orbitals are available, e.g., para-tricarbonylchromiumphenyl halides. The geometry of the S NAr transition state permits useful, qualitative conceptual distinctions to be made between this reaction and other modes of nucleophilic attack. We suggest that the leaving group order in the gas phase will be dependent on the exact combination of nucleophile, leaving group, and substrate framework. The element effect is not established for these reactions. An anionic nucleophile, thiolate, gives very early transition states and negative activation enthalpies with activated aryl halides. Nor has differential solvation of the different transition states been strongly emphasized. The polarity reversal, and hyperconjugative influences have received little or no attention in the past. In addition, a reversal of polarity of the C–X bond from reactant to transition state in the case of ArCl and ArBr compared to ArF also contributes to their difference in reactivity. Computational studies of the reaction of piperidine and dimethylamine with the same aryl halides using the polarizable continuum model (PCM) for solvation indicate that polar, polarizability, solvation, and negative hyperconjugative effects are all of some importance in producing the element effect in methanol. Experimental evidence shows that the element effect order in the reaction of piperidine with 2,4-dinitrophenyl halides in methanol is governed by the differences in enthalpies of activation. Multiple causes for this result have been proposed. The “element effect” in nucleophilic aromatic substitution reactions (S NAr) is characterized by the leaving group order, F > NO 2 > Cl ≈ Br > I, in activated aryl halides. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
